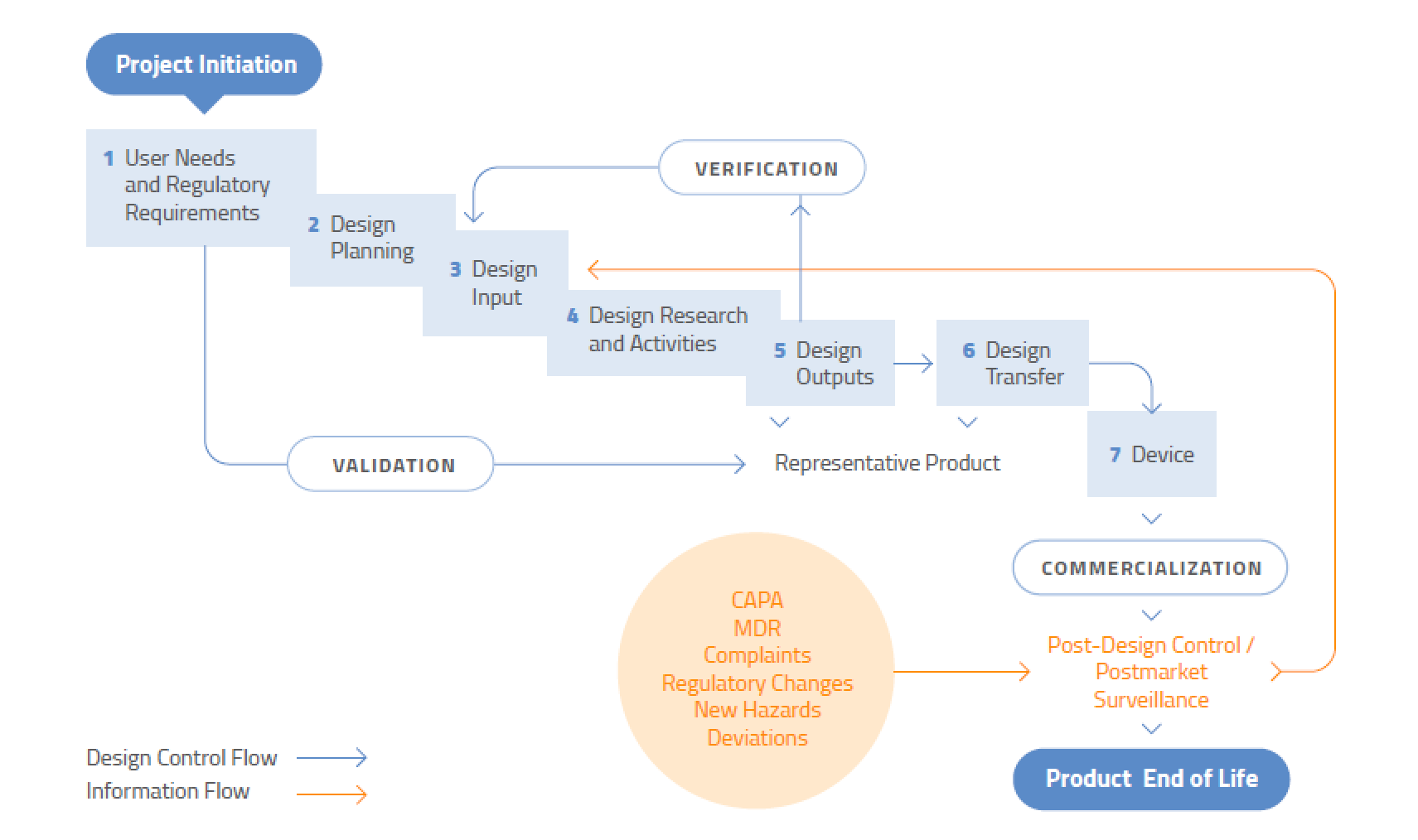
Revised text to reflect text in the medical devices regulations section 21 1 g updated reference to iso 8601 standard section 21 2 included reference to internet web address.
Medical device labeling standards.
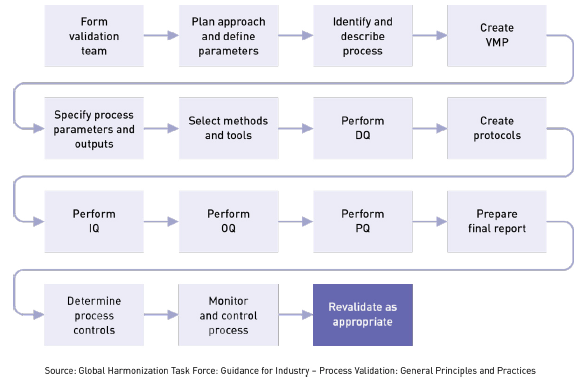
In iec 60601 1 labeling is deemed a critical component of a medical device 1 the standard provides comprehensive requirements for medical device marking and labeling.
Medical device labeling is considered as important as classifying a product or creating an insulation diagram.
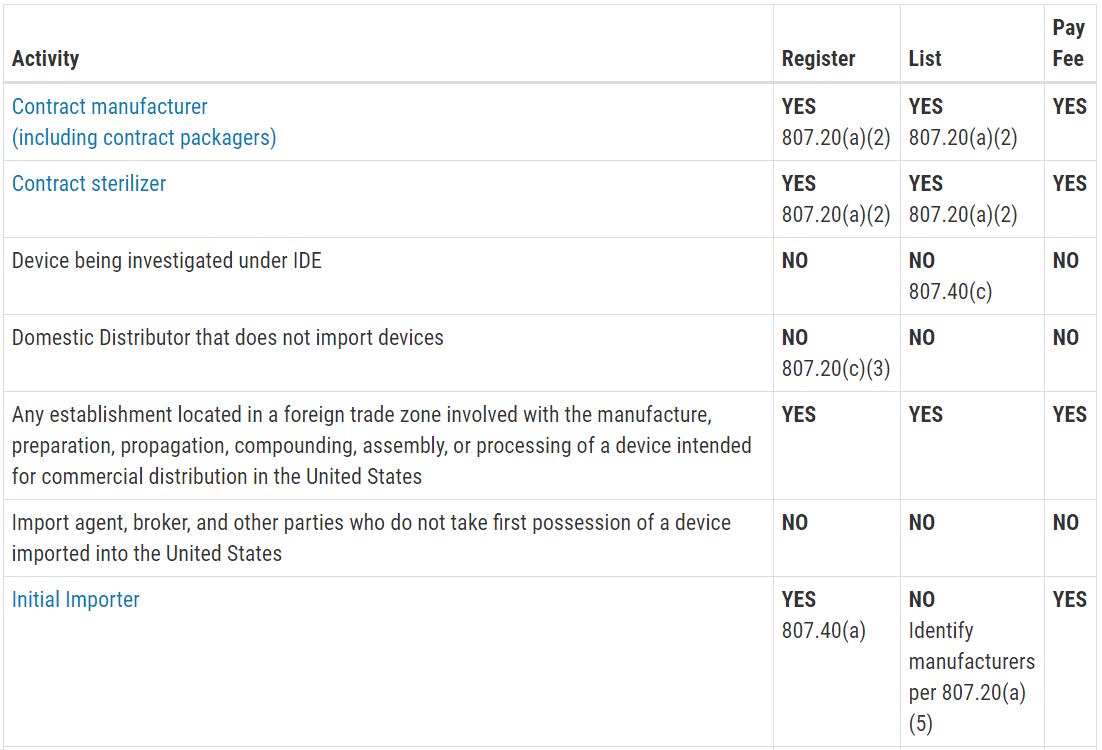
Labeling regulations pertaining to medical devices are found in the following parts of title 21 of the code of federal regulations cfr.
The general labeling requirements for medical devices are contained in 21 cfr part 801.
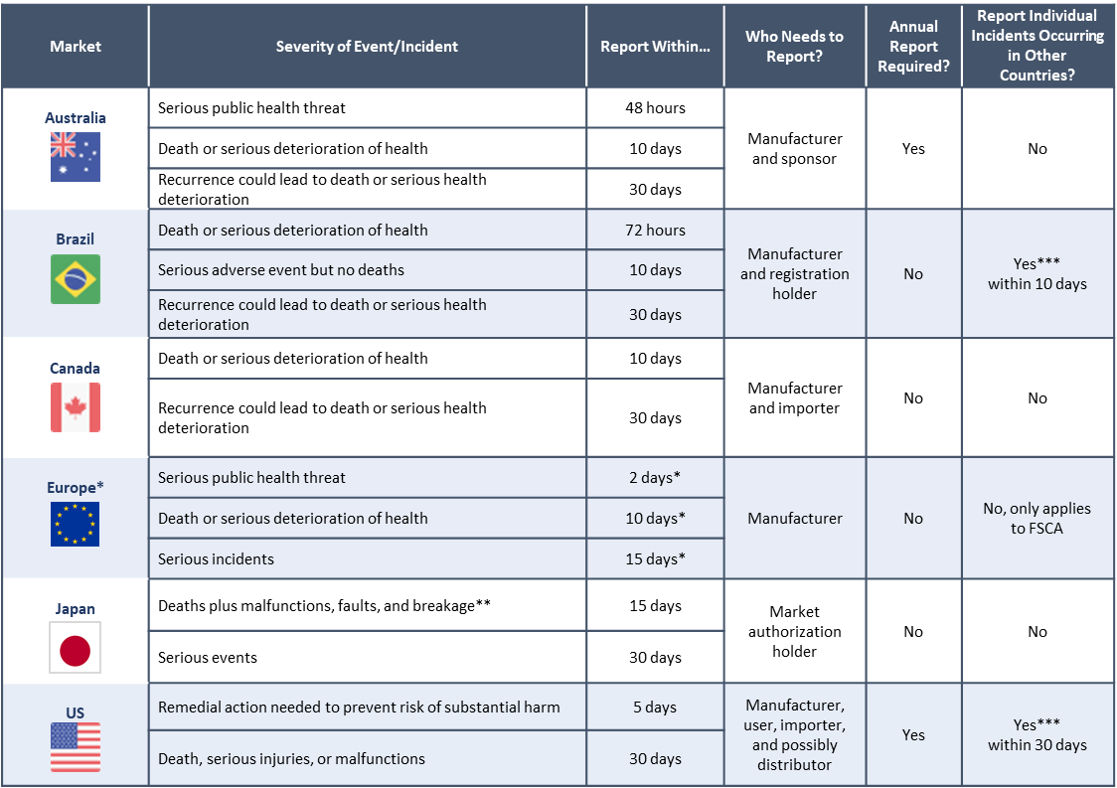
Essential principle 13 of schedule 1 of the therapeutic goods medical devices regulations 2002 the regulations outlines the requirements for information that must be provided.
An interlaboratory comparison of analytical methods for ethylene oxide pb 86.
801 127 medical devices.
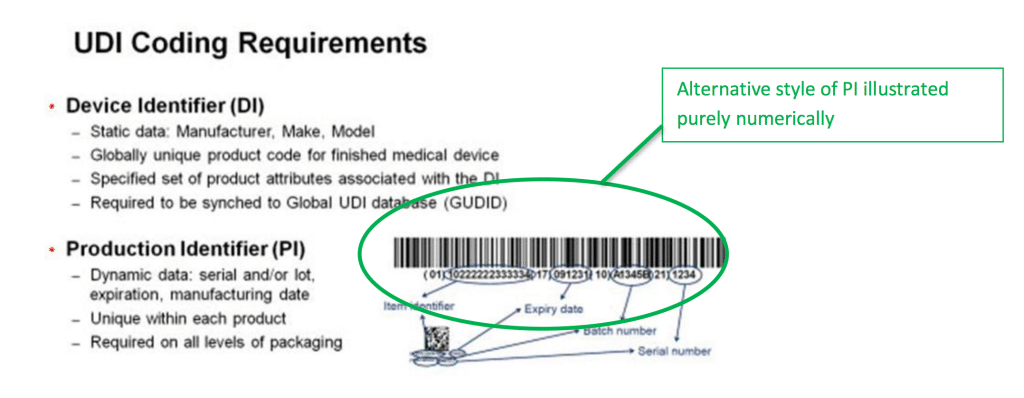
General device labeling 21 cfr part 801 use of symbols.
These regulations specify the minimum requirements for all devices.
Revised text for clarity included information on e labelling of certain medical devices not sold to the general public appendix 1.
All medical devices supplied in australia must meet the relevant essential principles for safety and performance to ensure the device is safe and performs as intended.
Device advice introduction to labeling requirements for medical devices including advertising over the counter exemptions in vitro diagnostics investigational devices quality system.
Labeling regulatory requirements for medical devices gpo 017 012 00327 3 2 75 pb 86 184348 as 11 95.
Iso 15223 1 2012 identifies requirements for symbols used in medical device labelling that convey information on the safe and effective use of medical devices.
It also lists symbols that satisfy the requirements of iso 15223 1 2012.
801 128 exceptions or alternatives to labeling requirements for medical devices held by the strategic national stockpile.

.jpg)